 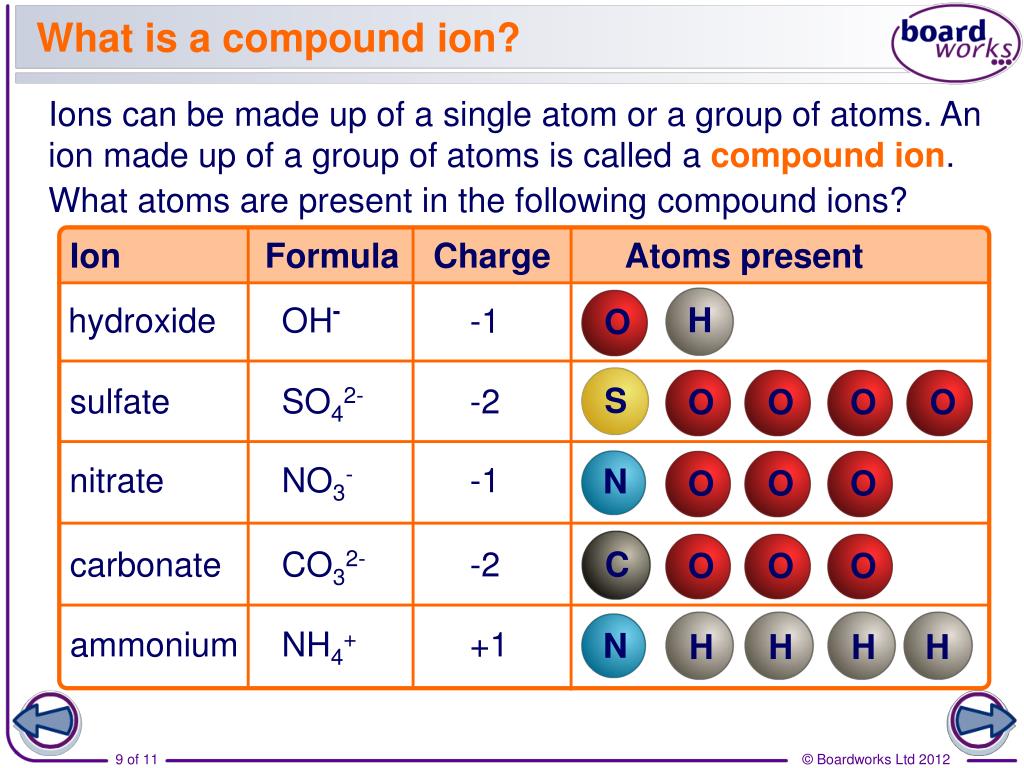
The video below shows how to use a technique called the crisscross method to determine the formula unit for an. So for every sulfide 2- ion there must be two silver 1+ ions. The overall charge of silver sulfide must be zero. Silver ions have a charge of 1+ and sulfide ions have a charge of 2. 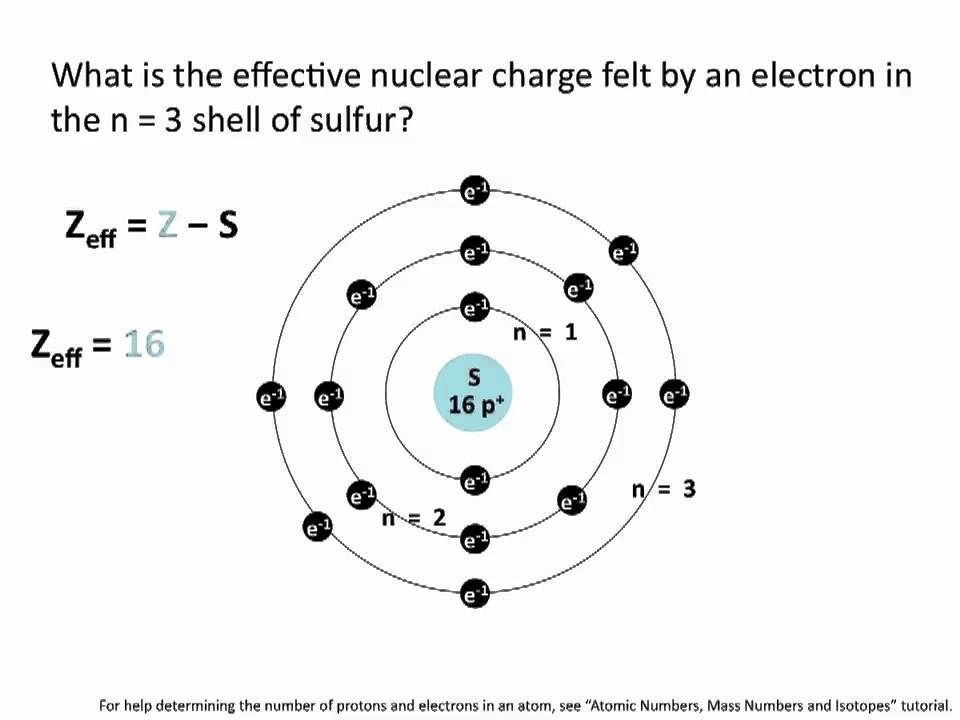
to balance it will need, one Ca 2+ ion and two OH - ions,ĭeduce the formula for sodium carbonate. The formula for silver sulfide is 'Ag'2'S'.Solubility product constant Ksp AgCl (s) Ag+ (aq) + Cl- (aq). this is two positive charges and one negative charge Anion, cation (common names, formulas and charges for familiar ions e.g., NH4+.ExampleĬalcium hydroxide contains Ca 2+ and OH - ions: If more than one polyatomic ion is needed, its formula is written inside brackets. The formulae of compounds containing polyatomic ions are worked out in a similar way to single atom ions. What Are Ions Properties of Matter Chemistry FuseSchoolWhat is an ion What role does it have to play in the structure of atoms Watch this video to fi. The table shows the names and formulae of some of these ions. Ionic Charges Chart Cations Roman numeral notation indicates charge of ion when element commonly forms more than one ion. But you need to know the specific ionic charge elments. Non-metals (found on the right) will be negative. On the Periodic Table metals (found on the left of the table) will be positive. To find the ionic charge of an element youll need to consult your Periodic Table. Polyatomic ions are formed from groups of atoms. Finding Charges of Ions on Periodic Table. The table shows the formulae of some common transition metal ions. The number in the name of the compound shows the charge of the metal ions in that compound.įor example, copper(II) sulfate contains Cu 2+ ions. Transition metals (in the central block between groups 2 and 3) can have ions with different charges. Exceptions: The transition metals Ag+1, Zn2+, and Cd2+ have fixed charges. Elements in the same group form ions with the same charge. Example: Al3+ is aluminum ion, Ga3+ is gallium ion. In some systems with high ion activity (e.g., saline soils, fertilizer bands), net adsorption may be limited by the capacity of the PRS® Probe to hold ions.The charge of many ions can be worked out using patterns in the periodic table. Conversely, declining activities of a strongly held ion may not result in the amount adsorbed by PRS® Probes unless the activity and affinity of other ions is sufficient to displace the strongly held ion. Increasing activity of an ion that is strongly held will generally increase the quantity of that ion on PRS® Probes and reduce the quantity of ions that are weakly held. The principles of ion exchange continue to apply. Soils are constantly undergoing change, and therefore the quantity of ions adsorbed on PRS® Probes may also change. 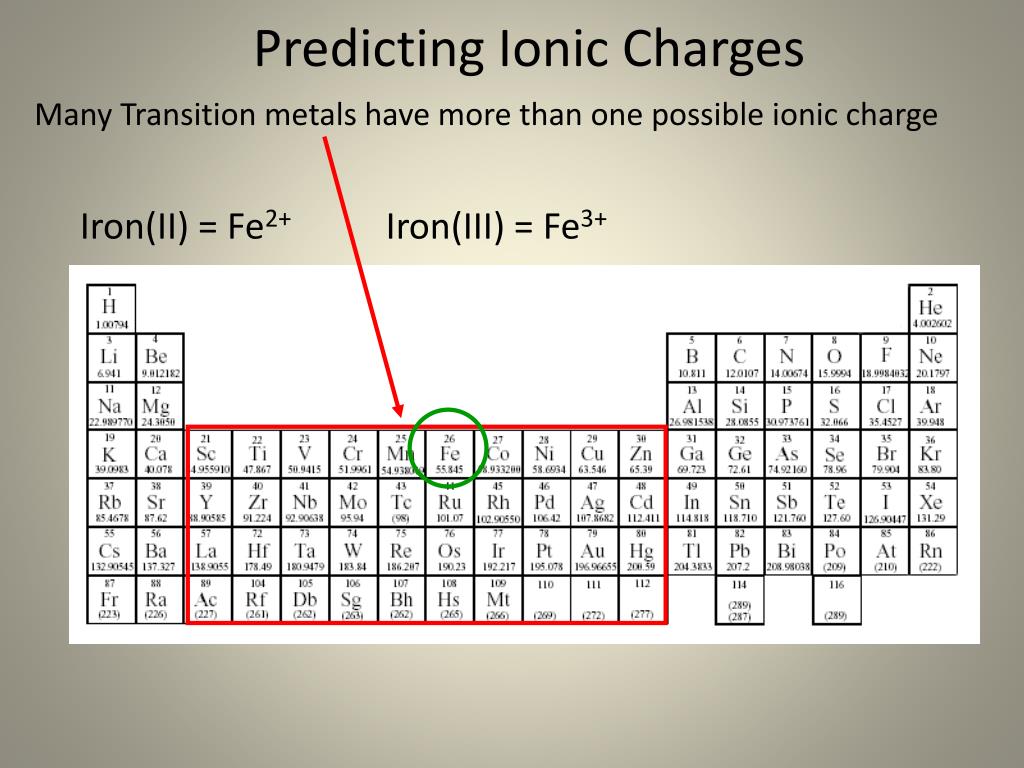
Metals generally have a tendency to lose one. Within a given valence, ions that have a smaller dehydrated radius are generally held more strongly. Silver (symbol: Ag) is a metal and has only 1 ion ( ), which is formed when the silver atom loses 1 electron. Relative affinity of an ion on a resin membrane (or other soil colloids) is most strongly influenced by valence: divalent ions are retained more strongly than monovalent ions, and trivalent ions are retained even more strongly. The recent advances in chloride-ion capturing electrodes for capacitive deionization (CDI) are limited by the capacity, rate, and stability of desalination. Un stockage sans entretien et une auto décharge quasi nulle - Les batteries AGM ont un taux d’autodécharge très faible, de l’ordre de 1 à 3 par mois. In both systems, a critical rate-limiting step is diffusion to the sink in response to gradients established by dynamic equilibria.Įventually, the quantity of a specific ion adsorbed on the ion exchange membrane often reaches a constant level that is Equilibrium Dependent, it depends on the relative activity in soil solution and relative affinity on the membrane of the ion. The process is similar to that occurring in the soil-root system, in which roots adsorb nutrients from soil solution by releasing counter ions such as H +, OH -, and HCO 3. When PRS® Probes are first inserted in soil, the rate of exchange is Diffusion Dependent: the flux of ions to the ion exchange membrane is controlled by the activity and diffusivity of ions in soil solution. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
